Sponsored by Nuvolo

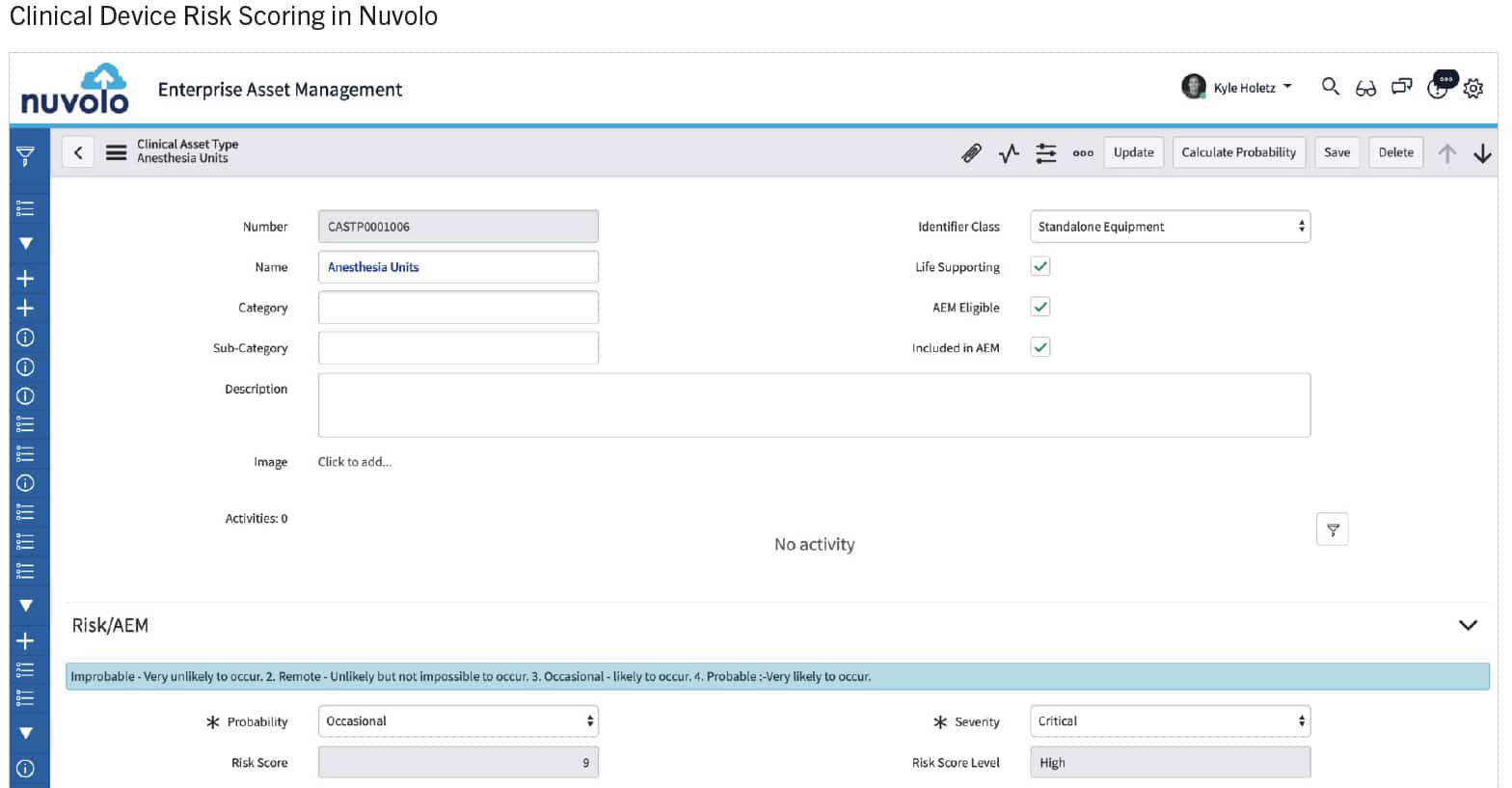
The Risk Scoring System for Medical Devices (RSS-MD) provides a methodology to catalog critical vulnerabilities and numerically score the potential severity of a failure with any clinical device. The Association for the Advancement of Medical Instrumentation (AAMI) published an Alternative Equipment Maintenance (AEM) guidebook in 2018 that recommended a risk scoring methodology that is now also being used to identify a medical device’s overall risk for potentially causing staff and patient harm. The two main components of this methodology include the probability of functional failure multiplied by the severity of the consequences for that harm. The scoring algorithm was developed using academic research principles in coordination with medical professionals and health care providers. The scoring algorithm uses a weighted scale to focus on the impact to patient safety, while incorporating the attributes of vulnerability characterization. The overall score (as shown in the chart below) provides a measurement that can be used for making Healthcare Technology Management (HTM) decisions and help prioritize mitigation efforts in a consistent and measurable manner for any hospital. These include determining corrective work order prioritization, hazard alert response, AEM eligibility and equipment replacement needs.
Risk Scoring Table

With so many medical equipment management decisions being based on this important safety metric, it’s necessary for HTM organizations to use a risk scoring method that not only accurately determines risk at a point in time, but tracks changes in risk over time. A recent TechNation webinar on June presented a model for health care organizations to standardize how they determine risk calculations and generate a universal understanding of the safety implication for different classes of clinical devices. This will help drive improvements for HTM audits and benchmarking of equipment performance.
The webinar covered the history of risk scoring for clinical devices along with definitions from both The Joint Commission (TJC) and the Centers for Medicare & Medicaid Services (CMS). Both have similar descriptions for risk scoring that underlines the importance of tracking high-risk devices. The TJC definition states, “Identify high-risk medical equipment for which there is a risk of serious injury or death to patient or staff should it fail.” In addition, the webinar described a necessary life-cycle approach to monitoring and managing the ongoing analysis and actions to control risk. This included the following process steps which should be made part of the computerized maintenance management system (CMMS) solution supported by the hospital HTM group:
- risk analysis
- risk evaluation
- risk control
- production and post-production information [analytics]
Risk Scoring for AEM Programs
The final topic discussed during the TechNation webinar was an overview of how risk scoring plays a key role in the allowance of AEM schedules. With some exceptions, an AEM program allows a health care facility to “adjust its maintenance, inspection, and testing frequency and activities for facility and medical equipment from what is recommended by the manufacturer, based on a risk-based assessment by qualified personnel” (CMS 2013). For many types of equipment, health care facilities find that the manufacturer-recommended maintenance activities are more onerous than is warranted, based on their own history of using and maintaining the device. This means that HTM groups can reduce (or increase if warranted) the number of maintenance checks and tasks required for clinical devices to save time, effort and the associated costs. These AEM schedules are authorized under TJC compliance models and can drive considerable cost savings for HTM teams.
However, to change the manufacturer-recommended maintenance requirements, the equipment must meet certain criteria and HTM organizations must adhere to specific guidelines. Therefore, tracking PM-related failures of equipment is very important. These should be monitored closely and allowed to trigger the removal of any device from an AEM schedule. Categories of these types of device failures include the following:
- Failures that could have been prevented by a better PM (Example: Early failure of infusion pump battery)
- Hidden failures that could have been discovered by a better PM (Example: Low defibrillator output)
Additional tracking recommended for risk scoring of devices on AEM schedules comes from the average, or mean time between failures (MTBF). This is calculated by using the model below:
For successful tracking and management of an AEM program, including risk scoring, a modernized CMMS solution is highly recommended to help organizations validate and manage the entire process, and to pass CMS accreditation surveys.
Conclusion
The Risk Scoring for Medical Devices was developed in response to gaps for evaluating associated risks of identified vulnerabilities in medical devices. The RSS-MD has been validated by a myriad of health care professionals and is currently being leveraged by major medical device manufacturers to assist in their risk assessment frameworks. The process of tracking and managing the life cycle of risk scoring for every clinical device is a daunting task. The more your CMMS can simplify the risk scoring calculations, data entry, tracking and management for your technicians, the more accurate your risk scoring data will be over time. Fortunately, newer, more nimble CMMS providers have emerged in recent years and are able to more easily configure their platforms to adhere to the changing regulations.
For more information about the clinical device risk scoring and AEM schedule management capabilities in Nuvolo, please reference our web page for clinical asset management at https://www.nuvolo.com/industry/healthcare.



