
By Ramya Kirhsnan
The procurement and deployment of a new patient monitoring system often introduces a significant and unwelcome clinical risk in the form of increased alarm fatigue. Beyond the operational burden of excessive noise, a high volume of non-actionable alerts erodes clinical trust in the technology and increases the likelihood of missed critical events.
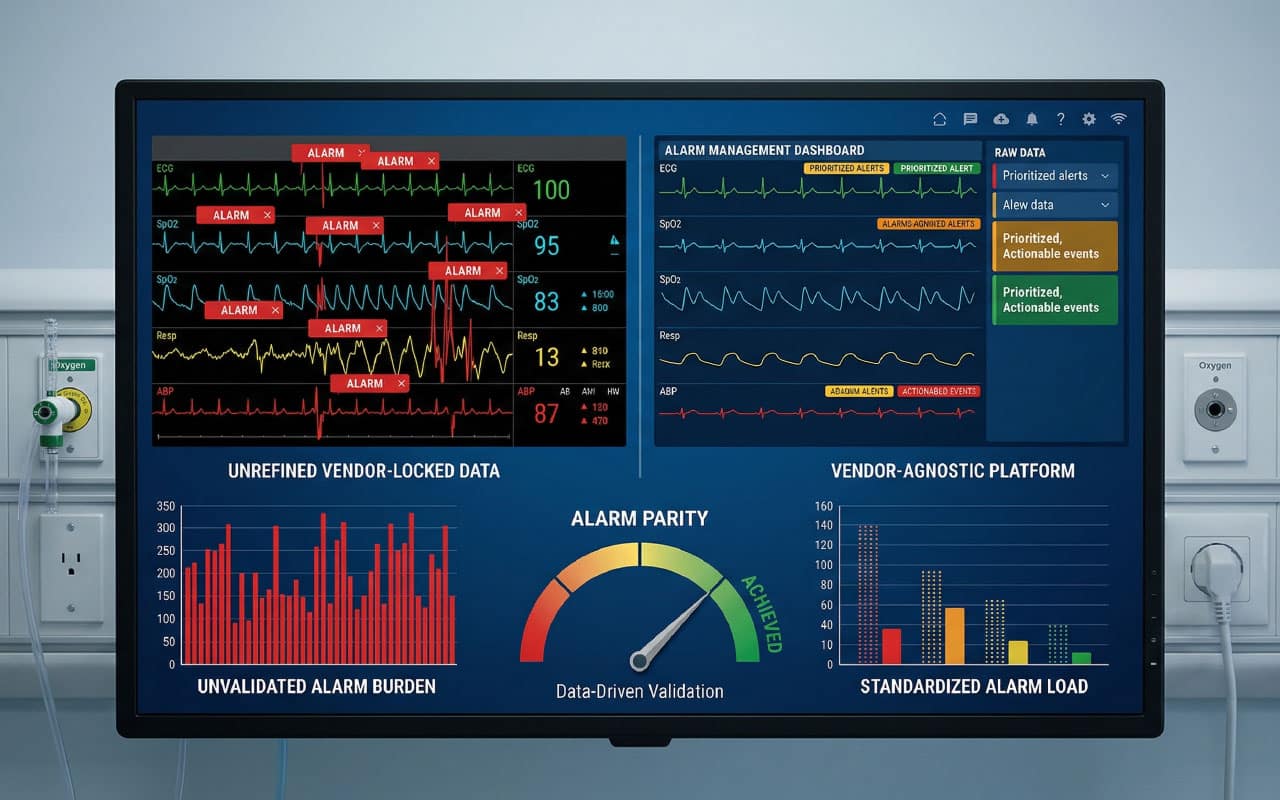
The fundamental issue is that different vendors employ proprietary backend logic and varying sensitivities that dictate how and when an alarm is triggered. Alarm parity, ensuring that a new system does not exceed the baseline alarm burden of the legacy platform, requires coordinated action by healthcare technology management (HTM) and clinical leadership through data-driven validation and integrated oversight.
INTEGRATED OVERSIGHT: A CLINICAL EVOLUTION
A successful transition is not a hardware swap; it is a clinical evolution. Critical to this process is a multidisciplinary task force comprising biomedical engineering, nursing leadership and IT. Biomedical engineering provides technical configuration expertise on the patient monitoring system, while nursing leadership offers the “on-the-ground” context necessary to validate the relevance of the changes to bedside workflows. In environments utilizing middleware, IT teams act as essential application owners overseeing the flow of data between disparate systems.
By embedding these teams into the project from the very beginning, the facility ensures that configuration changes are evidence-based and clinically relevant. This group must exist from the RFP phase through post-production to continuously monitor alarm loads and validate the effectiveness of implemented changes.
DATA COLLECTION: QUANTIFYING THE SCALE
Quantifying the scale and nature of the alarm problem is the first step to avoiding a “noise spike” at go-live. Gather data on the existing alarm burden to establish a baseline for the new system.
Ideally, to achieve an unbiased assessment, facilities should utilize third-party, vendor-agnostic, and alarm management platforms. These systems can enable a standardized, “apples-to-apples” comparison of raw data from both legacy and new vendors. It is important that the system provides a granular breakdown of alarm information (e.g., by alarm/device type) to help identify specific triggers – such as SpO2 thresholds or arrhythmia sensitivities – that may require adjustment. As an aside, these platforms can also typically gather alarm data from ancillary bedside devices (e.g., ventilators) to help with the larger alarm management effort.
In the absence of a vendor-agnostic alarm management platform, HTM teams can leverage the native reporting tools provided by the individual patient monitoring vendors. While these are often “vendor-locked,” comparing data from the legacy system against trial data from the new system can still provide a baseline to inform any configuration changes.
VALIDATION: THE PRODUCTION-GRADE “SANDBOX”
Data collection needs to be followed by validation in a controlled environment. During the validation process, the monitoring vendor should be expected to provide a test environment that mimics their production offering with recommended default configurations. This allows the multidisciplinary team to test the new vendor’s logic against known clinical scenarios without risking patient safety.
In an ideal world, facilities are able to create a robust test environment with the ability to mimic complex patient and alarm loads. Using a high-fidelity sandbox that replicates the full production alarm workflow across all vendors and systems, the team can evaluate configuration changes, including alarm delay and parameter averaging adjustments, to minimize nuisance alarms while maintaining clinically appropriate safety thresholds.
If a full-scale simulation is not feasible, at a minimum the team should simulate high-impact alarm scenarios (as identified by the clinical teams) on a subset of demo or test devices. By observing how the new hardware responds to specific physiological triggers, the team can identify any obvious differences between the new and legacy systems and account for them either by configuration changes or additional training before the fleet is rolled out to the floors.
CONCLUSION: ACHIEVING PARITY
The ultimate goal is “alarm parity” to ensure the new system does not exceed the alarm burden of the old one. By using a multidisciplinary team to drive data collection and rigorous pre-deployment testing, hospitals can move from reactive troubleshooting to proactive safety. Regardless of whether third-party middleware or foundational vendor reports are used, the strategy is consistent and requires validation of algorithms, default configurations, and the clinical environment prior to bedside deployment.
– Ramya Kirhsnan is director of implementations at Medical Informatics Corp. The opinions are those of the author.




