Today, the U.S. Food and Drug Administration (FDA) issued the draft guidance: “Predetermined Change Control Plans for Medical Devices.”
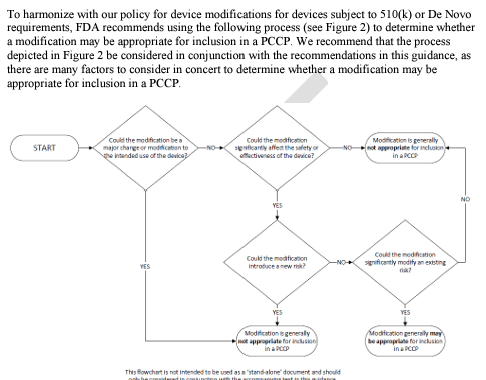
FDA is issuing this draft guidance to provide FDA’s current thinking on a policy for Predetermined Change Control Plans (PCCPs) and recommendations on the information to include in a PCCP in a marketing submission for a device. A PCCP is the documentation describing what modifications will be made to a device and how the modifications will be assessed. This draft guidance recommends that a PCCP for a device describe the planned device modifications, the associated methodology to develop, validate, and implement those modifications, and an assessment of the impact of those modifications. FDA reviews the PCCP as part of a marketing submission for a device to ensure the continued safety and effectiveness of the device without necessitating additional marketing submissions for implementing each modification described in the PCCP. The recommendations in this draft guidance apply to devices, including device-led combination products, reviewed through the 510(k), De Novo, and PMA pathways.
This draft guidance proposes a policy for predetermined change control plans (PCCPs) and recommendations on the information to include in a PCCP in a marketing submission for a device.
The FDA reviews the PCCP as part of a marketing submission for a device to ensure the continued safety and effectiveness of the device without needing to submit additional marketing submissions or obtain further FDA authorization before implementing each modification described in the PCCP.
Although you can comment on any guidance at any time (see 21 CFR 10.115(g)(5)), to ensure that the FDA considers your comment on a draft guidance before it begins work on the final version of the guidance, submit either online or written comments on the draft guidance before the close date.
If unable to submit comments online, please mail written comments to:
Dockets Management
Food and Drug Administration
5630 Fishers Lane, Rm 1061
Rockville, MD 20852
All written comments should be identified with this document’s docket number: FDA-2024-D-2338



