By K. Richard Douglas

In 1918, there was no such thing as a flu shot. The Spanish flu raged across the planet that year without hindrance. Those who contracted the disease could not be treated with a therapeutic because there were no antivirals at the time. A flu vaccine was not available in the U.S. until the 1940s.
The Spanish flu pandemic killed as much as three percent of the world’s population, including 675,000 Americans.
World War I lasted from 1914 to 1918. More U.S. soldiers died in 1918 from the Spanish flu than died in the entire war.
Not only was there no vaccine or therapeutics to fight the Spanish flu, but for those who worked in hospitals, there wasn’t the availability of PPE as we know it today. Compounding the problems for hospitals; the demands of the war created a shortage of physicians and other health care workers.
In the midst of the 2020 coronavirus pandemic, medical advances have helped physicians and researchers to more quickly understand this viral illness, yet because it is a new (novel) virus, it remains a vexing challenge.
A highly transmissible and deadly virus, that was first identified in the Wuhan Province of China, has proven that it can challenge any medical advances made since the 1918 pandemic.
The SARS-CoV-2 virus was first thought to be a more common virus causing pneumonia in patients. Physicians quickly learned that this new virus was both more deadly and more contagious than more common viruses like the flu virus.
As time went on, it became apparent to doctors and researchers that the new coronavirus not only caused pneumonia, but would block the alveoli in the lungs reducing the oxygen exchange in the lungs and the amount of oxygen traveling to organs. The new virus would also attack the heart, brain, liver and other organs in many patients leading to widespread inflammation and oxygen starvation. It can cause acute respiratory distress syndrome (ARDS).
As the immune system reacts to the virus’s attack, an over-reaction called a cytokine storm is possible.
For the most severely ill patients, the virus would cause multiple organ failure. Doctors were frustrated because few effective therapeutics were identified early on and the mortality rates in some places, like northern Italy, were extremely high.
A patient on a ventilator requires monitoring and care from several clinicians, so the demands put on staffing, along with clinical staff members who have become ill themselves, has made mitigation and treatment of the illness more difficult at many facilities.
The experience of the last major pandemic to hit the U.S. in 1918, which had real consequences and a substantial death toll, has been of little comfort to the biomed community. The experience of being thrown into a health care crisis, unlike anything seen in 102 years, requires tenacity, vigilance, empathy and ingenuity.
It has been in the face of this global emergency that HTM professionals have risen to the occasion and provided expertise and services that have saved lives and brought a degree of relief to their clinical colleagues.
Critical Equipment and Enough Beds
 HTM professionals have been faced with several similar challenges from the very onset of the crisis. The first was a need for specific equipment that was critical to the survival of the sickest patients.
HTM professionals have been faced with several similar challenges from the very onset of the crisis. The first was a need for specific equipment that was critical to the survival of the sickest patients.
In most cases, the existing inventories of these devices were wholly inadequate. Inventories were designed to accommodate the normal demands, or even a regional epidemic, but not a worldwide pandemic.
As part of the CARES Act, signed into law in March, 2020, the FDA was able to take steps to broaden access to diagnostic, therapeutic and protective medical devices by issuing Emergency Use Authorizations (EUAs) and dispensing a device shortage list.
Ventilators were the first devices that physicians demanded in abundance because SARS-CoV-2 was believed to be mostly a respiratory disease that had the potential to cause bilateral pneumonia. About five percent of COVID-19 patients require a ventilator.
State health departments and the federal government focused much of their efforts on these devices in response to the pleas of doctors.
The needs of any given hospital have been in constant flux, much of it based on how many mild, moderate, severe and critical COVID-19 patients are being treated on any given day. This requires surge planning that addresses any equipment needs forecast.
Device availability has not been the only area where biomeds have contributed to prepare their facilities for the influx of COVID-19 patients. There has also been the critical demand for special COVID-19 beds or rooms that had to be erected or stood-up quickly.
In some cases, this was merely an extension of an existing ICU and, in others, a retrofitting of a department with other safeguards and monitoring equipment was required and in other cases, a completely separate temporary unit had to be constructed.
Expanding hospital surge capacity meant cancelling elective surgeries to free up existing beds.
Many of these expansion sites, or CMS Hospitals Without Walls, allowed hospitals to construct additional capacity while CMS relaxed “certain conditions of participation (CoPs) and provider-based rules for hospital operations to maximize hospitals ability to focus on patient care,” according to the agency.
Addressing a surge of COVID-19 patients has also required needed equipment.
Invasive and non-invasive ventilators, along with consumables, accessories and spare parts, are a minimum requirement. An oxygen source is also required. The supply of accessories and consumables has to be adequately maintained.
With invasive ventilators, there is also a requirement for well-trained staff to maintain this equipment, as well as professionals capable of intubation. Both continuous positive airway pressure (CPAP) and bi-level positive airway pressure (BiPAP) devices were also identified early on as important devices to have on hand for COVID-19 patients who might not require a ventilator.
The other challenge has been the highly infectious or transmissible nature of the SARS-CoV2 virus. Not only were supplies of PPE in short supply during the early days of the pandemic, but the extra precautions to keep all staff safe, while monitoring patients, required additional precautions.
In many cases, to preserve the supply of PPE and to help protect clinicians, it was necessary to separate the monitoring equipment from the immediate vicinity of the patient. Biomeds sprang into action to distance ventilator monitors from patient beds.
Preparing the Facility
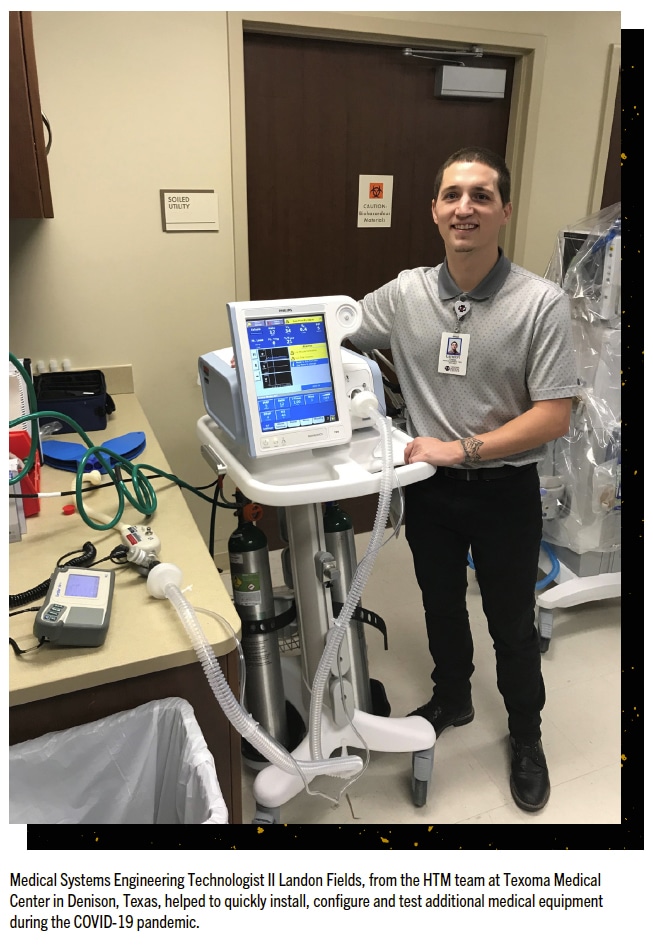 The need for a greater supply of ventilators has been a principal challenge for biomeds. Additionally, the need to install more monitoring capability has been another universal requirement in surge preparation.
The need for a greater supply of ventilators has been a principal challenge for biomeds. Additionally, the need to install more monitoring capability has been another universal requirement in surge preparation.
“During the COVID-19 pandemic at my facility, we had to quickly install additional patient monitors in an area that was converted to a COVID-only unit. Landon Fields, a Medical Systems Engineering Technologist II from our team stepped in to quickly install, configure, and test additional monitors and central stations allowing the nurses to constantly monitor the COVID patients,” says Ryan Harris, CBET, director of healthcare technology management at Texoma Medical Center in Denison, Texas.
Harris says that whenever COVID-19 required their patient care strategies to change, Landon was there at every turn to support the nursing staff in their equipment needs.
“Landon has also been very busy with keeping our ventilators in working condition during this pandemic by quickly repairing any that have problems. He has done an excellent job ensuring that the respiratory therapists have the equipment that they need during a time when having adequate ventilators is essential,” Harris says.
In Michigan, a large effort by a major health care system tackled the logistics and transport of critical equipment to address the COVID-surge needs in the community.
“Many sites such as McLaren Flint (Jeff Semple), McLaren Macomb (Scott Scandalito), McLaren Oakland (Tom Tesolin) and McLaren Northern Michigan (Bob Turk) all jumped to their emergency plans and their teams moved equipment such as monitors, beds, pumps and many other devices from closed sites such as outpatient surgery to expand the capability of inpatient units and build COVID-19 units,” says Samantha Jacques, Ph.D., FACHE, vice president at McLaren Clinical Engineering Services (MCES), at McLaren Health in Grand Blanc, Michigan.
Jacques says that her team set up a command center at their corporate clinical engineering building, where new purchases were unboxed and installed.
“All staff, including Gary Woods, who is our medical device integration specialist, unboxed and set up items such as high flow oxygen setups which were then dispersed to all our sites,” she says.
“We had a daily huddle with all sites to define local needs. We then transferred devices between sites including life-saving ventilators. Zella David, manager at McLaren Bay, drove ventilators from her site to other sites that were in need,” Jacques adds.
She says that Rick McCloy, the manager at McLaren Lansing, was responsible for testing utilization of ventilators for more than one patient.
“Although this option was never used, as it was a worst-case scenario, he defined the disposables and processes needed to use anesthesia machines to be used as well as how to modify a ventilator to be used on more than one patient,” Jacques says.
Standing Up Temporary Units
Capacity has been a challenge during the pandemic and biomed teams have been an invaluable resource to allow health care facilities to expand that capacity on short notice.
“In the earliest days of the pandemic we were called upon to provide equipment solutions and guidance on how to leverage our available resources to create surge capacity. Called upon to provide monitoring for our COVID cohort unit, they were able to redeploy a deinstalled telemetry system within 48 hours. Again, utilizing disused monitoring equipment they created a 15-bed monitored unit in a space that was repurposed in record time,” says Bob Meninno, director of biomedical engineering at Hoag Memorial Hospital Presbyterian Hospital in Newport Beach, California.

In places like Michigan and New York, facilities that were not designed to house medical patients had to be quickly transformed and retrofitted to address surge capacity.
“Detroit was a hot spot, so in addition to getting our hospitals ready, we also provided our clinical engineer, Pam Shuck, to a coalition of health care workers and emergency personnel who stood up the TCF center – an arena where the national guard and other emergency personnel built an additional care site. Pam was the only clinical engineer on the project and led the national guard staff in the acquisition and deployment of equipment as well as helped define some of the workflows for the clinical staff. She worked tirelessly more than 10 hours a day for weeks to prepare this site amid uncertainty and ambiguity,” Jacques says.
Facing Unique Challenges
Nations, governments and public health agencies, all attempt to prepare for pandemics and other public health emergencies. They provide guidance with suggestions for mitigating a pandemic. But, when the scope of the emergency is so pervasive that it overwhelms hospital ICU capacity, then health care professionals have to improvise and address challenges themselves.
That has been the case with many HTM departments.
“Our efforts and responsibilities have not abated past the initial preparedness push. The chronic lack of parts support continues. We have since secured dedicated warehouse space to allow us to stockpile both equipment and associated maintenance parts to buffer against the challenges,” Meninno says.
“We have been building our medical device resources, and in some cases, retaining older equipment we might otherwise have sold to avoid the abuses we had sometimes encountered when trying to obtain used equipment. Our contribution to the Hoag COVID Playbook has set forth a strategy to continuously improve on our independence from manufacturer support,” Meninno adds.
He says that his team, as part of this effort, has been building technical training into its capital process.
“Decisions on purchases are heavily weighted in favor of manufactures who choose to support us. We have maintained our compliance and quality measures despite our own staffing levels being impacted for a variety of reasons related to the pandemic,” Meninno says.
Another aspect of dealing with the coronavirus pandemic has been additional attention to protocols already in place in all hospitals. The concerns with disinfection become intensified when the pathogen in question cannot be addressed with antibiotics or other therapeutics.
“One of the things we have focused on, and continue to emphasize, is the disinfection of all medical equipment before it comes into the HTM department. Each of the techs has a disinfection station on their repair carts and there is a stop line before coming into the shop,” says Sterling Brown, CBET, unit director of healthcare technology management at Iredell Health System in Statesville, North Carolina.
“Although we assume that the clinical staff is wiping down/disinfecting equipment after it is discharged from a patient or prior to it being sent to the HTM department for servicing, we, as a matter of precaution wipe down every piece before we touch it or before it is allowed into the shop,” Brown adds.
He says that all of the techs have been fit tested for the N95 respirator and the powered air-purifying respirator (PAPR) so that they are protected if they are required to enter a COVID-positive patient room (and it happens on occasion).
“We vet all vendors that require access to the facility and we ensure they are temperature tested and have the proper PPE before entrance is allowed,” Brown says.
In Texas, one biomed team that is part of a large health care system, addressed the concern of equipment availability with resourceful ingenuity.
“The biomed team at BSW McKinney played an important role in helping the facility early during the pandemic. The first major role we played was repurposing and relocating equipment to be used in different departments and figuring out ways to make bedside monitors portable when all vendors were out of supplies. We converted old Welch Allyn BP roll stands, we had in storage, into roll stands for X2 modules and other patient monitors we previously did not have roll stands for,” says James Swandol, MBA, CBET, manager of healthcare technology management at Baylor Scott and White (BSW) McKinney.
He said that his team was also tasked to come up with a solution for patients to be able to speak to their families and physicians.
“We ended up mounting iPads onto roll stands so that patients could FaceTime family members from their room, and through different applications, the physicians could take advantage of telemedicine,” Swandol says.
Swandol says that gearing up for the surge was tough on the team.
“Ventilators were high priority on the list, during the first month we had a local college offer us loaner ventilators, which the team coordinated transportation of the equipment, and arranged for the equipment to be serviced by vendors. Since then, system solutions have been created to help every facility in our Baylor Scott and White system receive ventilators when they need them,” he says.
“When confronted with one vendor’s inability to provide parts, the team set about manufacturing required battery packs to support our transport ventilators. With the wealth of equipment-specific training our team possesses, we continued to provide support to the front line without requiring direct support for the OEMs, many of whom could not provide onsite support,” Meninno says.
Not only are patients and clinicians grateful for the valiant and persistent effort HTM professionals have made at their facilities, but HTM leadership has been reminded of their team’s important role.
“The biomedical engineering staff has been a truly wonderful resource for our organization. No one member shines above the rest, but rather they all excel together. I could not be prouder of them all,” Meninno says.
“It seemed like every day we were being tasked to come up with a solution to problems; huge shout out to the technicians who answered all calls to action to make sure we kept patients’ treatment and safety first,” Swandol says.
“It was a huge team effort and because of it no patient who needed a ventilator anywhere within the system didn’t have one,” Jacques says.
“To celebrate that effort, the Chamber of Commerce in Flint and Genesee County has named me as on the ‘20 in 20’ – the 20 people making a difference in our community during 2020. This is a huge honor and a great way to celebrate the clinical engineering field,” Jacques adds.
TechNation says thank you to the HTM heroes who have answered the call and saved countless lives when their skills and resourcefulness were needed most. The many victories during this pandemic can be attributed to your dedicated service.





